- admin
- 0 Comments
Could low-concentration atropine potentially replace orthokeratology as the leading method for myopia control in the future? I believe this is a topic that many doctors, patients, and parents are highly interested about.
I would like to share my views on this matter.
IN SHORT, HIGHLY UNLIKELY.
Below are my arguments:
Let’s first discuss the existing options that have been applied clinically in the field of myopia prevention and control, as well as those currently under development or close to clinical application. I will briefly elaborate on their clinical indications, the efficacy of myopia control, overall advantages, and limitations.
When mentioning the effectiveness of myopia control, I must first explain how efficacy is defined to ensure everyone's understanding.
The most commonly used definition for myopia control efficacy is the relative difference in myopia progression between the treatment group and the control group. Myopia progression can be evaluated based on changes in myopia and/or changes in axial length. For example, a 50% efficacy could mean that in the control group, children, on average, experience -1D of myopia progression per year, while in the treatment group, -0.5D/yr. Therefore, the efficacy is 50%.
Similarly, if axial elongation is used as the indicator, a 50% efficacy could mean that in the control group, children, on average, experience an axial elongation of 0.50mm/yr, while in the treatment group, 0.25mm/yr.
I want to emphasize here that the efficacy evaluated by these two indicators is not fully comparable.
1. Overnight Orthokeratology (OrthoK)
Orthok a type of specially designed gas- permeable rigid contact lenses that are worn overnight during sleep, and upon removal in the morning, clear vision without the need for glasses is achieved. OrthoK is FDA approved for the temporary correction of myopia. However, with their increasing use and growing patient population, clinical practitioners have gradually discovered that children undergoing Orthok experience a significantly slower progression of myopia compared to those wearing single vision eyeglasses
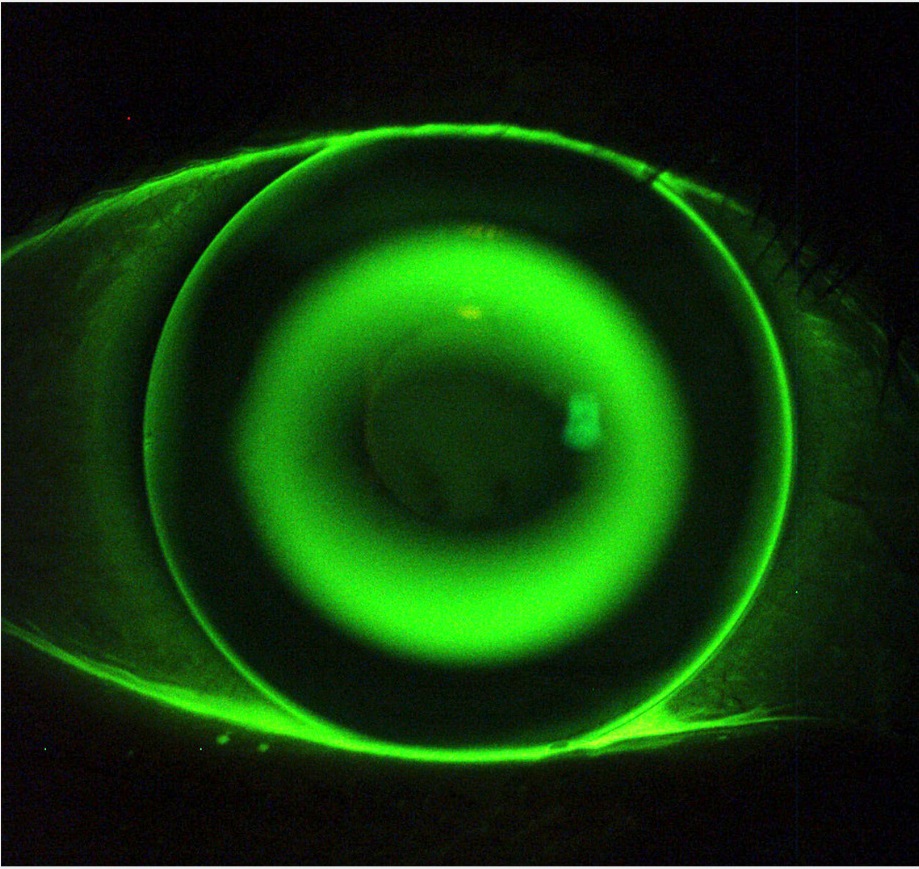
This clinical observation was subsequently confirmed in a series of clinical studies. Considering all the clinical research related to myopia control using Orthok, its long- term efficacy for myopia control, as measured by relative axial elongation, ranges from approximately 30% to 70%.
You might wonder, why is there such a significant difference in the efficacy individuals wearing Orthok?
The main reasons for significant individual differences are as follows:
1) each child has a different risk of rapid myopia progression. Generally, the earlier the age of myopia onset, the faster it progresses, and the more challenging it is to control;
2) the corneal surface is reshaped differently for each patient, and different corneal shapes correspond to different doses of myopia control;
3) one of the prerequisites for myopia control with Orthok is clear daytime vision after lens removal. For some patients who have poor daytime visual correction with the treatment, their myopia control effectiveness may also be affected.”
Nevertheless, due to its significant myopia control effectiveness and unique overnight wearing mode that offers convenience, Orthok has gradually become one of the preferred treatment options for a large number of myopic children.
Of course, orthokeratology is not a panacea; it has its limitations as well.
Firstly, to achieve the ideal fitting and ensure good daytime vision, as well as long-term safety. Orthok places high demands on the expertise of the doctor, the precision of the lens manufacturing, the ocular conditions of the patient (refractive error, corneal shape, eye health etc), and the compliance with wearing and care.
Secondly, the fitting and product cost of Orthok is not cheap, and it requires regular replacement. Therefore, the ideal population for the treatment is quite selective.
2. Daytime wear multifocal contact lens (MFCL)
MFCL have gradually become one of the main methods for myopia control internationally in recent years. MiSight 1d, one of the MFCL, has recently received approval from the U.S. FDA for myopia control. Based on existing clinical research data, the myopia control effect of multifocal contact lenses (evaluated by changes in refractive error and axial growth) is also around 30% to 70%
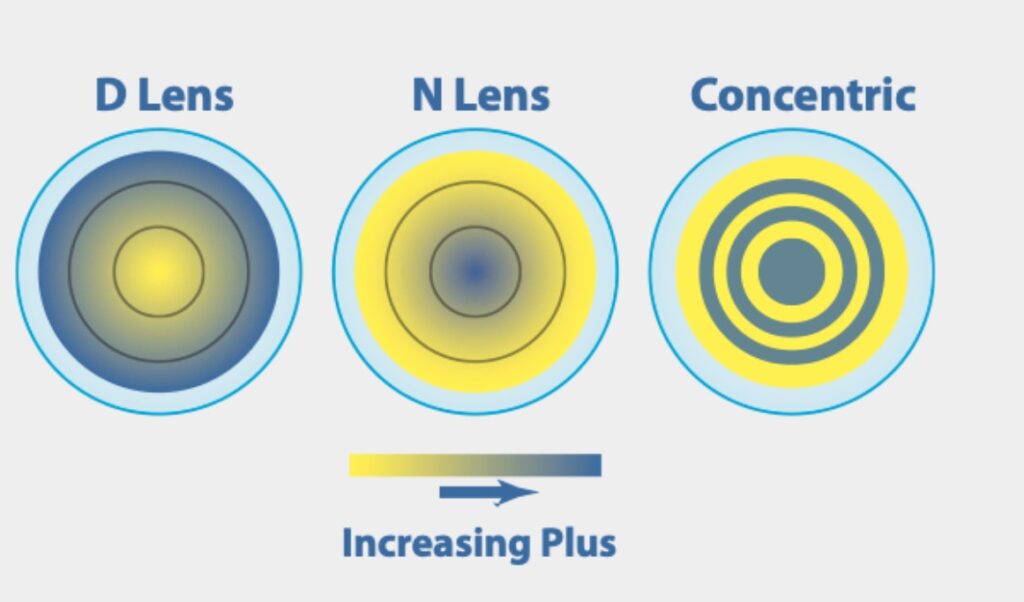
Currently, there is no clinical trial that directly compares the myopia control effects of MFCL and Orthok by randomly assigning participants to both groups simultaneously
Multifocal contact lenses (MFCL) complement orthokeratology (OK) lenses with the following three advantages
1) the fitting process is simpler, requiring less expertise from the doctor, and the number of visits is less frequent compared to Orthok treatment;
2) the soft lens replacement schedule can be more frequent (daily disposables, biweekly, or monthly disposables);
3) there are virtually no restrictions on the level of myopia to be considered for MFCL option.
Limitations of MFCL
Firstly, clear vision relies on wearing the lenses during the day, so there is a higher requirement for wearers to independently put on and take off the lenses; and it is not ideal for water activities.
Secondly, due to its multifocal optical design, the corrected vision by MFCL may be slightly weaker than with single-vision lenses.
Thirdly, the cost of the lenses is higher, and for daily disposal option, the annual cost is higher than OrthoK treatment.
3. Low dose atropine (LDA) eye drop
Atropine is commonly used as ophthalmic diagnostic and therapeutic eye drops. Although at higher concentrations (0.5-1%), atropine can cause significant photophobia and blurred vision, its lower concentrations for long-term use before bedtime show significantly improved tolerance.

The long-term efficacy and safety of low concentration (0.025~0.05%) atropine for myopia control have been increasingly supported by numerous clinical studies, including large-sample placebo-controlled randomized clinical trials, internationally. Currently, several multicenter pre-market clinical trials are also underway, with the hope of obtaining the US FDA approval for myopia control around 2025.
It is worth noting that the
preparation of low-concentration atropine eye drops is very complex
The long-term safety and efficacy of continuous use depend not only on the precise control of the concentration of the active ingredient but also on the selection and concentration of preservatives, as well as the rational design of non-active ingredients (such as solvent medium, buffering salts, additives, isotonic agents, etc),
The efficacy of atropine in controlling myopia, as well as the incidence and severity of its side effects, and the tolerability of treatment, all show a clear dose-response relationship. The higher the concentration used, the more significant the myopia control effect (20%-80%), but at the same time, the side effects become more pronounced, and long- term use becomes less tolerable.
Furthermore, the higher the concentration used, the more significant the rebound effect caused by sudden discontinuation (i.e., a significant acceleration of myopia progression after stopping treatment). Therefore, how to determine the initial concentration of atropine use, indications for increasing concentration, and the tapering schedule before discontinuation to achieve a balance between control efficacy and safety/tolerability requires more clinical trial data to support.
Nevertheless, due to its convenience of use, relatively simple exam process, and lack of restriction on patients’ refractive errors, the use of low-concentration atropine for myopia control is increasingly accepted by a wide range of optometrists and ophthalmologists.
The main limitation of LDA lies in its singular focus on myopia control, while lacking myopia correction benefit
Therefore, patients using LDA still need to wear other optical corrections during the day to achieve clear vision. This significant limitation implies that LDA cannot replace the use of Orthok or MFCL in patients who strongly desire spectacle independence, but it is more likely to serve as a supplementary (combined) approach alongside the former two
Summary
Under the guidance of evidence-based medicine, the three methods of myopia control – OrthoK, MFCL, and LDA – all have their pros and cons, and they target different groups of patients. Currently, there is no clear evidence to show the superiority or inferiority of a specific method in myopia control. Therefore, factors such as the understanding of myopia prevention and control by medical professionals, product selection, patients’ lifestyles and visual habits, self- awareness and biases towards different methods, and parents’ predispositions will still be the dominant factors in the final choice.
It is important to emphasize that myopia prevention and control go beyond simply controlling the level of myopia and axial elongation or reducing the risk of myopia- related eye complications. It involves multiple considerations, including the perception of treatment benefit, cultural differences, and the self-esteem of myopic children. Predicting the future market of relevant methods and products solely based on myopia control efficacy is one-sided and impractical.
With a deeper understanding of the pathogenesis and prevention mechanisms of myopia, the future of myopia prevention and control will be personalized and tailored to each patient’s risk factors, clinical manifestations, and specific needs. It will revolve around integrated medical services combining optical, pharmaceutical, and behavioral approaches, with product offerings serving as supplementary tools in a comprehensive management approach.
In the “martial arts” of myopia prevention and control, there won’t be a single ultimate sword to dominate the scene.
